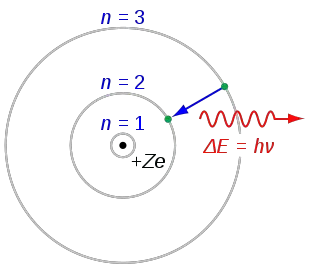
Modern atomic theory is not based on these old concepts. This ancient idea was based in philosophical reasoning rather than scientific reasoning. The word atom is derived from the ancient Greek word atomos, which means "uncuttable". The basic idea that matter is made up of tiny indivisible particles is an old idea that appeared in many ancient cultures. Chemistry is the science that studies these changes. The ability of atoms to attach and detach from each other is responsible for most of the physical changes observed in nature. This is a form of nuclear decay.Ītoms can attach to one or more other atoms by chemical bonds to form chemical compounds such as molecules or crystals. In this case, the nucleus splits and leaves behind different elements. Under certain circumstances, the repelling electromagnetic force becomes stronger than the nuclear force. This force is usually stronger than the electromagnetic force that repels the positively charged protons from one another. The protons and neutrons in the nucleus are attracted to each other by the nuclear force. The electrons of an atom are attracted to the protons in an atomic nucleus by the electromagnetic force. Conversely, if it has more protons than electrons, it has a positive charge, and is called a positive ion (or cation). If an atom has more electrons than protons, then it has an overall negative charge, and is called a negative ion (or anion). If the numbers of protons and electrons are equal, as they normally are, then the atom is electrically neutral as a whole. The electrons are negatively charged, and this opposing charge is what binds them to the nucleus. Protons have a positive electric charge and neutrons have no charge, so the nucleus is positively charged. More than 99.94% of an atom's mass is in the nucleus. Atoms are so small that accurately predicting their behavior using classical physics is not possible due to quantum effects. This is smaller than the shortest wavelength of visible light, which means humans cannot see atoms with conventional microscopes. A human hair is about a million carbon atoms wide. Atoms with the same number of protons but a different number of neutrons are called isotopes of the same element.Ītoms are extremely small, typically around 100 picometers across. For example, any atom that contains 11 protons is sodium, and any atom that contains 29 protons is copper. The chemical elements are distinguished from each other by the number of protons that are in their atoms. An atom consists of a nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The atom is the basic particle of the chemical elements. Smallest recognized division of a chemical elementĮlectrons and a compact nucleus of protons and neutrons The black bar is one angstrom ( 10 −10 m or 100 pm). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. Electrons do not significantly contribute to the mass of an atom.An illustration of the helium atom, depicting the nucleus (pink) and the electron cloud distribution (black). They play a critical role in determining the stability and mass of the nucleus.Įlectrons are subatomic particles with a negative charge and are found outside the nucleus of an atom in regions called electron shells or orbitals. Neutrons are subatomic particles with no electric charge and are found in the nucleus of an atom along with protons. It also contributes to the mass of the atom. They determine the atomic number of an element, which is the number of protons in the nucleus. Protons are positively charged subatomic particles found in the nucleus of an atom. The K shell is the first energy level closest to the nucleus and can hold a maximum of 2 electrons. This model helps us understand basic atomic properties and electron behavior in atoms. Electrons can jump between these energy levels by absorbing or emitting energy. The Bohr model describes the structure of an atom as a central nucleus containing protons and neutrons, with electrons orbiting in specific energy levels around it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
